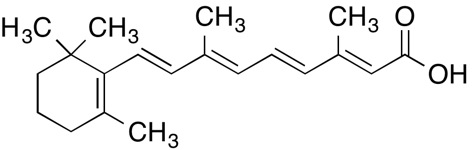
INCI name: retinoic acid
Synonyms: tretinoin, all-trans-retinoic acid, vitamin A acid
Molecular formula: C20H28O2
Molecular weight: 300.44 g/mol
IUPAC name:
(2E,4E,6E,8E)-3,7-dimethyl-9-(2,6,6-trimethylcyclohexen-1-yl)nona-2,4,6,8-tetraenoic acid
CAS number: 302-79-4
Retinoic acid is the carboxylic acid form of retinol (vitamin A). Retinal is the aldehyde analog of the molecule and is needed by the retina of the eye for vision. The retinoids play an important role in biology, often regulating key pathways in the body as well as controlling differentiation, proliferation, and apoptosis. Retinoids are commonly used to treat acne vulgaris or photodamage skin. Most of the present knowledge of the mechanism and function of trans-retinoic acid in skin comes from studies of the epidermis. It binds to retinoic acid and retinoic X receptors triggering a series of biochemical events ultimately stimulating keratinocyte proliferation and increasing the thickness of the epidermal layer. In addition, retinoids affect the dermis by such actions on fibroblasts as the down regulation of collagenase (an enzyme that breaks down peptide bonds in collagen), which would ultimately lead to damaged structural collagen fibrils in the dermis and the alteration of skin mechanical properties and visual aesthetics.
Properties1
Melting point: 180-182 °C
Solubility: Insoluble in water; slightly soluble in chloroform and ethanol; barely soluble in ether; soluble in methylene chloride.
Stability: The solution form of retinoic acid is sensitive to light, heat, and air.
References
1.
Sigma product information sheet; Sigma Prod. No. R2625 (2016).