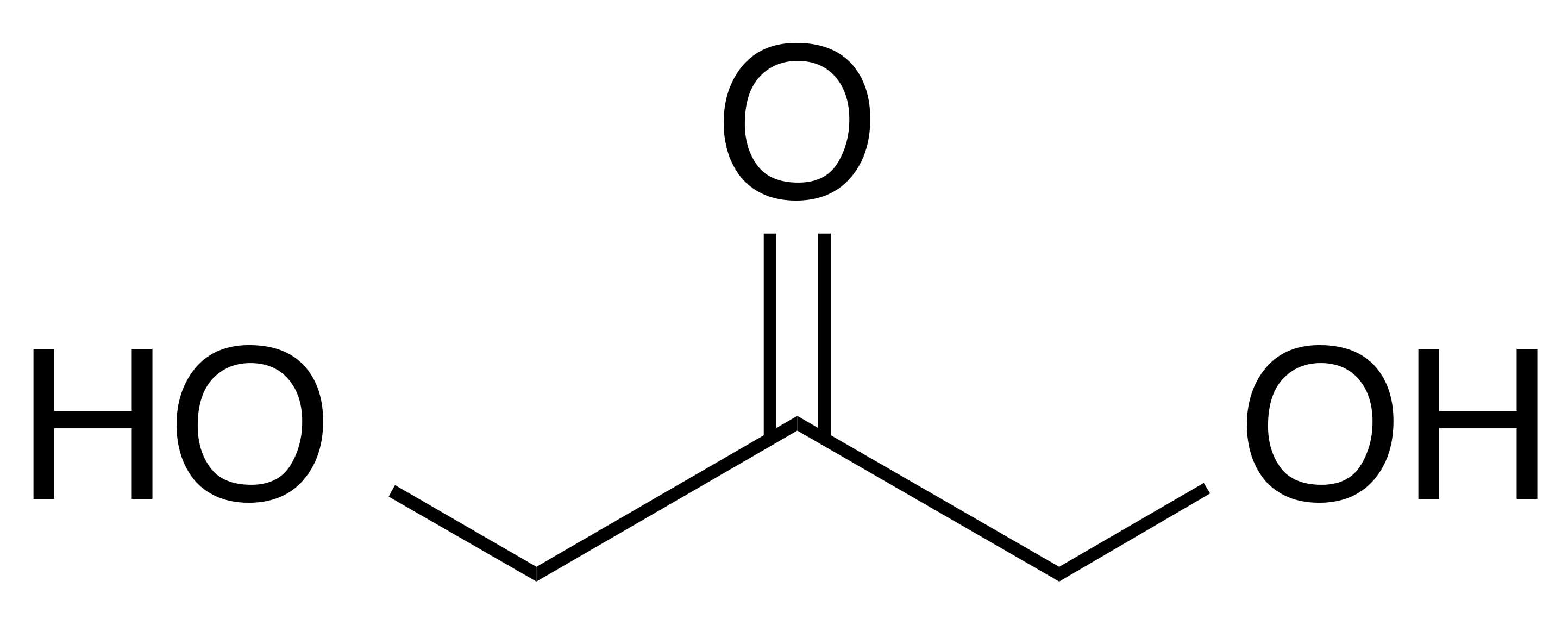
INCI name: dihydroxyacetone
Synonyms: 1,3-dihydroxyacetone
Molecular formula: C3H6O3
Molecular weight: 90.08 g/mol
IUPAC name: 1,3-dihydroxypropan-2-one
CAS number: 96-26-4
EC number: 202-494-5
Dihydroxyacetone is a self-tanning agent that when applied to skin is believed to undergo the Maillard reaction, resulting in the formation of a brownish species in the stratum corneum in the skin. The Maillard reaction typically occurs between glycosidic hydroxyl groups of saccharides and the amino group of amino acids and proteins. Dihydroxyacetone, in its dimeric form, adapts the structure of a monosaccharide. Since the reaction is limited to the stratum corneum, it is a semi-permanent effect, which subsides as the skin is exfoliated.
Publications on the effect of dihydroxyacetone on the skin date back to the early 1960s.1,2 Nevertheless, there has been considerable interest, even in recent years, in elucidating some of the properties of dihydroxyacetone and its biochemical mechanism of action in skin. There has been some controversy in the literature as it has been reported that dihydroxyacetone can cause DNA damage in cell culture studies involving keratinocytes and melanocytes.3,4 It is also been suggested to penetrate beyond the stratum corneum into the viable epidermis.3 Further, it has been demonstrated that dihydroxyacetone results in the formation of free radicals in skin in the presence of UV radiation.5
On the other hand, studies have shown the potential for dihydroxyacetone as a protecting agent of the skin from solar radiation.6,7 It is very important that we keep in mind that, in the United States, dihydroxyacetone is not regulated as a sunscreen, and is classified as a cosmetic.8 Nevertheless, there is tremendous interest in better understanding the properties of dihydroxyacetone as a self-tanning agent, especially with heightened consumer awareness of the perils of solar radiation exposure.9
Properties
Melting point: 75-80 °C10
Solubility: soluble in water and ethanol10
References
1. E. Wittgenstein and G.M. Guest, Biochemical effects of dihydroxyacetone, J. Invest. Dermatol., 37, 421-426 (1961).
2. E. Wittgenstein and H.K. Berry, Staining of skin with dihydroxyacetone, Science, 132, 894-895 (1960).
3. K.R. Smith, M. Granberry, M.C.B. Tan, C.L. Daniel, and N.R. Gassman, Dihydroxyacetone induces G2/M arrest and apoptotic cell death in A375P melanoma cells, Environ. Toxicol., 33, 333-342 (2018).
4. A.B. Petersen, H.C. Wulf, R. Gniadecki, and B. Gajkowska, Dihydroxyacetone, the active browning ingredient in sunless tanning lotions, induces DNA damage, cell-cycle block and apoptosis in cultured HaCaT keratinocytes, Mutat. Res. Genet. Toxicol. Environ. Mutagen., 560, 173-186 (2004).
5. K. Jung, M. Seifert, T. Herrling, and J. Fuchs, UV-generated free radicals (FR) in skin: their prevention by sunscreens and their induction by self-tanning agents, Spectrochim. Acta A Mol. Biomol. Spectrosc., 69, 1423-1428 (2008).
6. N. Rajatanavin, S. Suwanachote, and S. Kulkollakarn, Dihydroxyacetone: a safe camouflaging option in vitiligo, Int. J. Dermatol., 47, 402-406 (2008).
7. A.B. Petersen, R. Na, and H.C. Wulf, Sunless skin tanning with dihydroxyacetone delays broad-spectrum ultraviolet photocarcinogenesis in hairless mice, Mutat. Res. Genet. Toxicol. Environ. Mutagen., 542, 129-138 (2003).
8. T.L. Braunberger, A.F. Nahhas, L.M. Katz, N. Sadrieh, and H.W. Lim, Dihydroxyacetone: a review, J. Drugs Dermatol., 17, 387-391 (2018).
9. I. Sole, S. Vilchez, J. Miras, N. Montanya, M.J. Garcia-Celma, and J. Esquena, DHA and L-carnitine loaded chitosan hydrogels as delivery systems for topical applications, Colloids Surf. A, 525, 85-92 (2017).
10. M.J. O’Neil, The Merck Index, 15th ed., The Royal Society of Chemistry: Cambridge, UK (2013).